Some forms of energy which can be converted into heat energy are as follows:
- Mechanical Energy—this includes all methods of producing increased motion of molecules such as friction, impact of bodies, or compression of gases.
- Electrical Energy—electrical energy is converted to heat energy when an electric current flows through any form of resistance such as an electric iron, electric light, or an electric blanket.
- Chemical Energy—most forms of chemical reaction convert stored potential energy into heat. Some examples are the explosive effects of gunpowder, the burning of oil or wood, and the combining of oxygen and grease.
- Radiant Energy—electromagnetic waves of certain frequencies produce heat when they are absorbed by the bodies they strike such as x-rays, light rays, and infrared rays.
- Nuclear Energy—energy stored in the nucleus of atoms is released during the process of nuclear fission in a nuclear reactor or atomic explosion.
- Sun—all heat energy can be directly or indirectly traced to the nuclear reactions occurring in the sun.
When a gas is compressed, work is done and the gas becomes warm or hot. Conversely, when a gas under high pressure is allowed to expand, the expanding gas becomes cool. In the first case, work was converted into energy in the form of heat; in the second case heat energy was expended. Since heat is given off or absorbed, there must be a relationship between heat energy and work. Also, when two surfaces are rubbed together, the friction develops heat. However, work was required to cause the heat, and by experimentation, it has been shown that the work required and the amount of heat produced by friction is proportional. Thus, heat can be regarded as a form of energy.
According to this theory of heat as a form of energy, the molecules, atoms, and electrons in all bodies are in a continual state of motion. In a hot body, these small particles possess relatively large amounts of kinetic energy, but in cooler bodies they have less. Because the small particles are given motion, and hence kinetic energy, work must be done to slide one body over the other. Mechanical energy apparently is transformed, and what we know as heat is really kinetic energy of the small molecular subdivisions of matter.
Heat Energy Units
Two different units are used to express quantities of heat energy. They are the calorie and the BTU. One calorie is equal to the amount of heat required to change the temperature of 1 gram of water 1 degree Centigrade.This term “calorie” (spelled with a lower case c) is 1/1,000 of the Calorie (spelled with a capital C) used in the measurement of the heat energy in foods. One BTU is defined as the amount of heat required to change the temperature of 1 lb of water 1 degree Fahrenheit (1 °F). The calorie and the gram are seldom used in discussing aviation maintenance. The BTU, however, is commonly referred to in discussions of engine thermal efficiencies and the heat content of aviation fuel.
A device known as the calorimeter is used to measure quantities of heat energy. In application, it may be used to determine the quantity of heat energy available in 1 pound of aviation gasoline. A given weight of the fuel is burned in the calorimeter, and the heat energy is absorbed by a large quantity of water. From the weight of the water and the increase in its temperature, it is possible to compute the heat yield of the fuel. A definite relationship exists between heat and mechanical energy. This relationship has been established and verified by many experiments which show that:
One BTU of heat energy = 778 ft-lb of work
As discussed earlier in the Energy post under the topic “Potential Energy,” one pound of aviation gasoline contains 18,900 BTU of heat energy. Since each BTU is capable of 778 ft-lb of work, 1 lb of aviation gasoline is capable of 14,704,200 ft-lb of work.
Heat Energy and Thermal Efficiency
Thermal efficiency is the relationship between the potential for power contained in a specific heat source, and how much usable power is created when that heat source is used. The formula for calculating thermal efficiency is:Thermal Efficiency = Horsepower Produced ÷ Potential Horsepower in Fuel
For example, consider the piston engine used in a small general aviation airplane, which typically consumes 0.5 lb of fuel per hour for each horsepower it creates. Imagine that the engine is creating 200 hp. If we multiply 0.5 by the horsepower of 200, we find the engine is consuming 100 lb of fuel per hour, or 1.67 lb per minute. Earlier in this site, one horsepower was found to be 33,000 ft-lb of work per minute.
The potential horsepower in the fuel burned for this example engine would be:
Hp = 1.67 lb/minute × 18,900 BTU/lb × 778 ft lb/BTU
33,000 ft-lb/min
Hp = 744
The example engine is burning enough fuel that it has the potential to create 744 horsepower, but it is only creating 200. The thermal efficiency of the engine would be:
Thermal Efficiency = Hp Produced ÷ Hp in Fuel
= 200 ÷ 744
= .2688 or 26.88%
More than 70 percent of the energy in the fuel is not being used to create usable horsepower. The wasted energy is in the form of friction and heat. A tremendous amount of heat is given up to the atmosphere and not used inside the engine to create power.
Heat Transfer
There are three methods by which heat is transferred from one location to another or from one substance to another. These three methods are conduction, convection, and radiation.Conduction
Heat transfer always takes place by areas of high heat energy migrating to areas of low heat energy. Heat transfer by conduction requires that there be physical contact between an object that has a large amount of heat energy and one that has a smaller amount of heat energy.Everyone knows from experience that the metal handle of a heated pan can burn the hand. A plastic or wood handle, however, remains relatively cool even though it is in direct contact with the pan. The metal transmits the heat more easily than the wood because it is a better conductor of heat. Different materials conduct heat at different rates. Some metals are much better conductors of heat than others. Aluminum and copper are used in pots and pans because they conduct heat very rapidly. Woods and plastics are used for handles because they conduct heat very slowly.
Figure 1 illustrates the different rates of conduction of various metals. Of those listed, silver is the best conductor and lead is the poorest. As mentioned previously, copper and aluminum are used in pots and pans because they are good conductors. It is interesting to note that silver, copper, and aluminum are also excellent conductors of electricity.
 |
| Figure 1. Conductivity of various metals |
Liquids are poorer conductors of heat than metals. Notice that the ice in the test tube shown in Figure 2 is not melting rapidly even though the water at the top is boiling. The water conducts heat so poorly that not enough heat reaches the ice to melt it.
 |
| Figure 2. Water as a poor conductor |
Gases are even poorer conductors of heat than liquids. It is possible to stand quite close to a stove without being burned because air is such a poor conductor. Since conduction is a process whereby the increase in molecular energy is passed along by actual contact, gases are poor conductors.
At the point of application of the heat source, the molecules become violently agitated. These molecules strike adjacent molecules causing them to become agitated. This process continues until the heat energy is distributed evenly throughout the substance. The gases are much poorer conductors of heat because molecules are farther apart in gases than in solids.
Materials that are poor conductors are used to prevent the transfer of heat and are called heat insulators. A wooden handle on a pot or a soldering iron serves as a heat insulator. Certain materials, such as finely spun glass or asbestos, are particularly poor heat conductors. These materials are therefore used for many types of insulation.
Convection
Convection is the process by which heat is transferred by movement of a heated fluid (gas or liquid). For example, an incandescent light bulb will, when heated, become increasingly hotter until the air surrounding it begins to move. The motion of the air is upward. This upward motion of the heated air carries the heat away from the hot light bulb by convection. Transfer of heat by convection may be hastened by using a ventilating fan to move the air surrounding a hot object. The rate of cooling of a hot electronics component, such as the CPU in a computer, can be increased if it is provided with copper fins that conduct heat away from the hot surface. The fins provide large surfaces against which cool air can be blown.A convection process may take place in a liquid as well as in a gas. A good example of this is a pan of water sitting on the stove. The bottom of the pan becomes hot because it conducts heat from the surface it is in contact with. The water on the bottom of the pan also heats up because of conduction. As the heated water starts to rise and cooler water moves in to take its place, the convection process begins.
When the circulation of gas or liquid is not rapid enough to remove sufficient heat, fans or pumps are used to accelerate the motion of the cooling material. In some installations, pumps are used to circulate water or oil to help cool large equipment. In airborne installations, electric fans and blowers are used to aid convection.
An aircraft air-cooled piston engine is a good example of convection being used to transfer heat. The engine shown in Figure 3 is a Continental IO-520, with six heavily finned air-cooled cylinders. This engine does not depend on natural convection for cooling, but rather forced air convection coming from the propeller on the engine. The heat generated inside the engine finds its way to the cylinder cooling fins by conduction, meaning transfer within the metal of the cylinder. Once the heat gets to the fins, forced air flowing around the cylinders carries the heat away.
 |
| Figure 3. Aircraft piston engine cooled by convection |
Radiation
Conduction and convection cannot wholly account for some of the phenomena associated with heat transfer. For example, the heat one feels when sitting in front of an open fire cannot be transferred by convection because the air currents are moving toward the fire. It cannot be transferred through conduction because the conductivity of the air is very small, and the cooler currents of air moving toward the fire would more than overcome the transfer of heat outward. Therefore, there must be some way for heat to travel across space other than by conduction and convection.The existence of another process of heat transfer is still more evident when the heat from the sun is considered. Since conduction and convection take place only through some medium, such as a gas or a liquid, heat from the sun must reach the earth by another method, since space is an almost perfect vacuum. Radiation is the name given to this third method of heat transfer.
The term “radiation” refers to the continual emission of energy from the surface of all bodies. This energy is known as “radiant energy.” It is in the form of electromagnetic waves, radio waves, or x-rays, which are all alike except for a difference in wave length. These waves travel at the velocity of light and are transmitted through a vacuum more easily than through air because air absorbs some of them. Most forms of energy can be traced back to the energy of sunlight. Sunlight is a form of radiant heat energy that travels through space to reach the earth. These electromagnetic heat waves are absorbed when they come in contact with nontransparent bodies. The result is that the motion of the molecules in the body is increased as indicated by an increase in the temperature of the body.
The differences between conduction, convection, and radiation may now be considered. First, although conduction and convection are extremely slow, radiation takes place at the speed of light. This fact is evident at the time of an eclipse of the sun when the shutting off of the heat from the sun takes place at the same time as the shutting off of the light. Second, radiant heat may pass through a medium without heating it. In application, the air inside a greenhouse may be much warmer than the glass through which the sun’s rays pass. Third, although heat transfer by conduction or convection may travel in roundabout routes, radiant heat always travels in a straight line. For example, radiation can be cut off with a screen placed between the source of heat and the body to be protected.
Specific Heat
One important way in which substances differ is in the requirement of different quantities of heat to produce the same temperature change in a given mass of the substance. Each substance requires a quantity of heat, called its specific heat capacity, to increase the temperature of a unit of its mass 1 °C. The specific heat of a substance is the ratio of its specific heat capacity to the specific heat capacity of water. Specific heat is expressed as a number which, because it is a ratio, has no units and applies to both the English and the metric systems.It is fortunate that water has a high specific heat capacity. The larger bodies of water on the earth keep the air and solid matter on or near the surface of the earth at a constant temperature. A great quantity of heat is required to change the temperature of a large lake or river. Therefore, when the temperature falls below that of such bodies of water, they give off large quantities of heat. This process keeps the atmospheric temperature at the surface of the earth from changing rapidly.
The specific heat values of some common materials are listed in Figure 4.
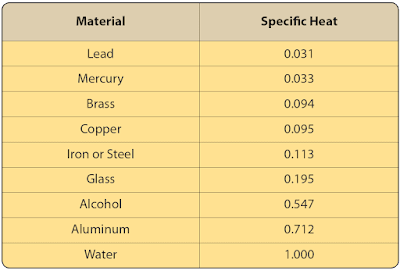 |
| Figure 4. Specific heat value for various substances |
Temperature
Temperature is a dominant factor affecting the physical properties of fluids. It is of particular concern when calculating changes in the state of gases.The three temperature scales used extensively are the Centigrade, the Fahrenheit, and the absolute or Kelvin scales. The Centigrade scale is constructed by using the freezing and boiling points of water, under standard conditions, as fixed points of zero and 100, respectively, with 100 equal divisions between. The Fahrenheit scale uses 32° as the freezing point of water and 212° as the boiling point, and has 180 equal divisions between. The absolute or Kelvin scale is constructed with its zero point established as minus 273 °C, meaning 273° below the freezing point of water. The relationships of the other fixed points of the scales are shown in Figure 5.
 |
| Figure 5. Comparison of temperature scales |
When working with temperatures, always make sure which system of measurement is being used and know how to convert from one to another. The conversion formulas are as follows:
Degrees Fahrenheit = (1.8 × Degrees Celsius) + 32
Degrees Celsius = (Degrees Fahrenheit – 32) × 5⁄9
Degrees Kelvin = Degrees Celsius + 273
Degrees Rankine = Degrees Fahrenheit + 460
For purposes of calculations, the Rankine scale is commonly used to convert Fahrenheit to absolute. For Fahrenheit readings above zero, 460° is added. Thus, 72 °F equals 460° plus 72°, or 532° absolute. If the Fahrenheit reading is below zero, it is subtracted from 460°. Thus −40 °F equals 460° minus 40°, or 420° absolute. It should be stressed that the Rankine scale does not indicate absolute temperature readings in accordance with the Kelvin scale, but these conversions may be used for the calculations of changes in the state of gases.
The Kelvin and Centigrade scales are used more extensively in scientific work; therefore, some technical manuals may use these scales in giving directions and operating instructions. The Fahrenheit scale is commonly used in the United States, and most people are familiar with it. Therefore, the Fahrenheit scale is used in most areas of this site.
Thermal Expansion/Contraction
Thermal expansion takes place in solids, liquids, and gases when they are heated. With few exceptions, solids will expand when heated and contract when cooled. The expansion of solids when heated is very slight in comparison to the expansion in liquids and gases because the molecules of solids are much closer together and are more strongly attracted to each other. The expansion of fluids is discussed in the study of Boyle’s law. Thermal expansion in solids must be explained in some detail because of its close relationship to aircraft metals and materials.It is necessary to measure experimentally the exact rate of expansion of each one because some substances expand more than others. The amount that a unit length of any substance expands for a one degree rise in temperature is known as the coefficient of linear expansion for that substance. The coefficient of linear expansion for various materials is shown in Figure 6.
 |
| Figure 6. Coefficient of expansion for various materials |
To estimate the expansion of any object, such as a steel rail, it is necessary to know three things about it: its length, the rise in temperature to which it is subjected, and its coefficient of expansion. This relationship is expressed by the equation:
Expansion = (coefficient) × (length) × (rise in temperature)
If a steel rod measures exactly 9 ft at 21 °C, what is its length at 55 °C? The coefficient of expansion for steel is 11 × 10−6.
Expansion = (11 × 10−6) × (9 feet) × 34°
Expansion = 0.003366 feet
This amount, when added to the original length of the rod, makes the rod 9.003366 ft long. Its length has only increased by 4⁄100 of an inch.
The increase in the length of the rod is relatively small, but if the rod were placed where it could not expand freely, there would be a tremendous force exerted due to thermal expansion. Thus, thermal expansion must be taken into consideration when designing airframes, power plants, or related equipment.
RELATED POSTS